Jacqueline Collins

Audio By Carbonatix
When it comes to marijuana concentrate, the question of “How much is too much?” won’t just apply to the product anymore. In Colorado, it will soon apply to the packaging.
A law mandating new packaging and labeling for extracted THC products is still in the early stages, with the state Marijuana Enforcement Division given until November to come up with ways to create more public education about the health risks of concentrated THC products, as well as visual changes to packaging to reflect serving sizes. The law, House Bill 1317, brings a long list of new rules and restrictions to the state’s medical marijuana program and creates state programs that will research THC’s effects on mental health.
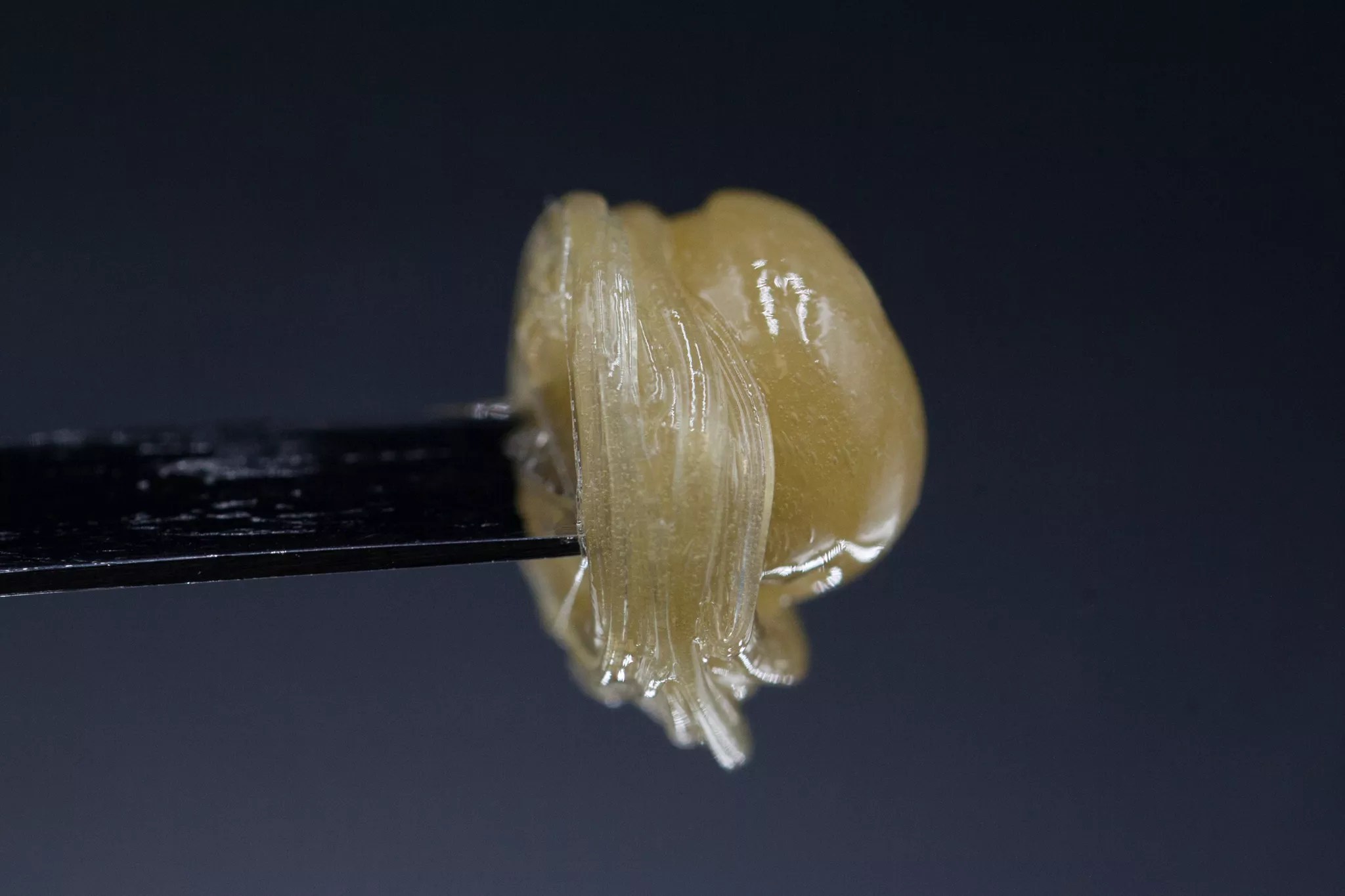
However, the law also demands that the MED craft new packaging and labeling rules for all marijuana concentrates, medical and recreational, enforcing new warnings and a standard serving size for wax, shatter, bubble hash, kief, live resin, vaporizer cartridges and all other extracted THC products. But not all THC concentrates are created equally, nor are they consumed in the same way. Kief, for instance, is powder-like and tests at around 25 to 30 percent THC; in contrast, wax and shatter reach around 70 percent THC and are sticky but solid substances. Live resin or THC crystalline can surpass 80 percent THC and has a more oily, liquid structure.
“I think the first step of the question is determining what that recommended serving size dose is going to be, because that visual representation will likely differ by product and by route of administration,” Dr. Jesse Hinkley, a psychiatry professor at the University of Colorado School of Medicine, said during a MED rulemaking session on August 11
“The variability of products, the variability of potency, the desire to protect costs and profit margins, is not a reason to not have a standard or equivalent dose across products,” he continued.
The bill originally proposed that all concentrated THC products be individually packaged in servings of ten per gram, similar to the way that edibles are currently packaged. After pushback from the pot industry, however, that language was replaced with a directive for the MED to oversee the creation of new packaging rules, which will take effect in 2022. Since then, basing the servings on milligrams of THC or size – such as the size of a grain of rice or sand – have both been suggested, but the argument still centers on dosage.
Proponents of HB 1317 want concentrate to be packaged or visually represented in 5- or 10-milligram servings of THC, but marijuana extractors say that would be too costly; other pot industry representatives warned against standardizing doses that could be larger than a new user expects.
“Not only is that against the statutory intent, but it’s going to make the cost of these products a lot more. You’re turning a $30 dollar concentrate into a $120 concentrate just based on the packaging and labor,” argued Kevin Gallagher, executive director of the Colorado Cannabis Manufacturers Association.
The visualization aspect is just part of the challenge for the MED and its rotating board of stakeholders. New educational materials for dispensaries and extra warning labels on concentrates are also mandated in HB 1317 – but what, exactly, customers should be warned about also came under debate. All medical and recreational marijuana products already bear warnings about the long-term physical and mental health risks of marijuana, as well as warnings for those about to drive or pregnant and breastfeeding mothers, but the point of HB 1317 was to get more specific.
Hinkley and other bill supporters on the panel pushed for warnings about suicide, psychosis and even self-harm from accidental burning, as the majority of THC concentrate consumption involves heating metal or glass to extremely high temperatures. Martha Montemayor, director of medical marijuana health-care group Cannabis Clinicians Colorado, suggested adding information about ways to curb the intoxicating effects of THC, citing citicoline and olivetol as potential remedies that are readily accessible – but she also warned against putting too much on a label in fear of losing attention spans.
“We seem to be heading toward the giant foldout thing on your prescription drugs that nobody reads. Let’s make sure we don’t head there,” Montemayor said during the meeting.
There was an agreement that any sort of educational information or potential labeling should include information on how to access professional help for mental health issues, and the possibility of a QR code for access to information online remains a possibility, though that would likely be supplemental. The next rulemaking meeting for concentrates is set for Friday, August 20, with the new rules taking effect on January 1, 2022.