YouTube

Audio By Carbonatix
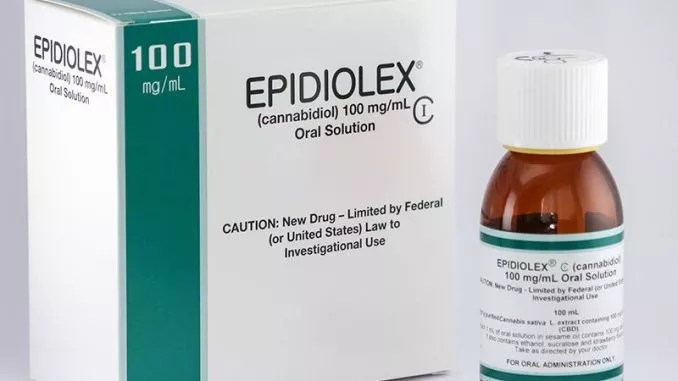
The Drug Enforcement Administration appeared to take a large step forward on Thursday, September 27, when it confirmed that it would reclassify Epidiolex as a Schedule V substance. The move follows Food and Drug Administration approval and classifies the marijuana-derived cannabidiol (CBD) medication under the DEA’s lowest restriction for drugs, so physicians and pharmacies can now prescribe and dispense it in all fifty states under federal law.
Despite the headlining news, the reaction in Colorado was a mixed bag, ranging from ho-hum to angry disappointment.
National organizations such as the National Organization for the Reform of Marijuana Laws responded with cautious optimism while reminding us of the high price of Epidiolex, a drug made by GW Pharmaceuticals used to treat Dravet syndrome and Lennox-Gastaut syndrome, two rare forms of epilepsy that cause seizures.
“It remains to be seen to what degree physicians will be comfortable prescribing this new agent and whether most patients can feasibly afford it,” NORML deputy director Paul Armentano says. “We anticipate that Epidiolex will be the first of many potential FDA-approved medicines based on the cannabis plant. These are welcome alternatives. But these products should not be regulated in such a manner that patients no longer have ready access to herbal cannabis.”
According to a report from the Wall Street Journal in August, GW Pharma plans to charge $32,500 annually per patient for Epidiolex, which could cost anywhere from $5 to $200 per month depending on the patient’s insurance.
Because medical and adult-use marijuana are both legal in Colorado, the General Assembly was able to prepare in case the DEA did not reclassify the medication. In April, the legislature passed HB 1187 to allow doctors to prescribe Epidiolex upon the FDA’s approval – which ended up coming in June – creating protections for health-care professionals similar to those around synthetic THC medication, like Marinol. In May, the legislature also passed HB 1295, a law that now protects hemp products from being deemed “adulterated” by the Colorado Food and Drug Act, making hemp products fit for human consumption under state law.
Still, the DEA’s recent move created waves in Colorado’s CBD industry, and one prominent cannabis law firm is pissed off.
The Denver-based Hoban Law Group represents clients in the marijuana, hemp and CBD industries, and the firm believes the DEA is issuing a reclassification that disrespects CBD’s current legal status.
“In re-scheduling Epidiolex, DEA overbroadly references not only Epidiolex but additionally any CBD product ‘derived from cannabis.’ Yet again, DEA fails to distinguish such products derived from lawful hemp as being exempted from DEA’s authority and the [Controlled Substances Act],” the law firm writes in response to the rescheduling.
Find the DEA’s summary on Epidiolex’s reclassification below:
With the issuance of this final order, the Acting Administrator of the Drug Enforcement Administration places certain drug products that have been approved by the Food and Drug Administration (FDA) and which contain cannabidiol (CBD) in schedule V of the Controlled Substances Act (CSA). Specifically, this order places FDA-approved drugs that contain CBD derived from cannabis and no more than 0.1 percent tetrahydrocannabinols in schedule V. This action is required to satisfy the responsibility of the Acting Administrator under the CSA to place a drug in the schedule he deems most appropriate to carry out United States obligations under the Single Convention on Narcotic Drugs, 1961. Also consistent therewith, DEA is adding such drugs to the list of substances that may only be imported or exported pursuant to a permit.
Industrial hemp plants can produce more CBD than psychoactive marijuana and must contain less than 0.3 percent THC to be considered hemp under federal law. Hoban believes that 2014’s Farm Bill allows states to legalize industrial hemp and hemp products and ship those products across state lines. But language in the DEA’s recent decision doesn’t differentiate between psychoactive marijuana and hemp (both plants are technically cannabis), and it says that only FDA-approved CBD medication would receive the Schedule V status.
“Given DEA’s tendency to misinterpret and ignore legislation, judicial decisions and its own directive regarding hemp, our firm will swiftly evaluate all available options and remedies for judicial action as in prior HIA v. DEA cases as well as administrative action through notice-and-comment on this new, misguided DEA action,” managing partner Bob Hoban says in a statement.
The DEA did not immediately respond to requests for comment. In June, the agency clarified that not all cannabinoids fall under the Controlled Substances Act, referencing a 2004 decision by the Ninth Circuit Court of Appeals that excluded non-psychoactive cannabinoids from the federal government’s definition of marijuana.
Morris Beegle, president of the Colorado Hemp Company and founder of the NoCo Hemp Expo (which just expanded to Nashville), believes the recent DEA decision confuses the market, calling the reclassification an “ambiguous” move. Still, he wasn’t as moved by the decision as Hoban.
“Most hemp people expected this,” Beegle says. “It’s specifically for the pharmaceutical side; it’s marijuana-derived. So I think the position of our industry is that this has nothing really to do with hemp-derived CBD. The hemp-derived CBD market is really putting out a general wellness product that could help with a variety of issues.”